 You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. Iron has an electron configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6. The number of valence electrons is 3, but they include electrons in a p-type orbital, so to calculate the group number we need to add 10 to the number of valence electrons. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). The highest occupied electron shell is 2, so the period number is 2. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. Thus, it is convenient to separate electrons into two groups. Copper and chromium have one valence electron (they are exceptions), because they have one 4s electron. 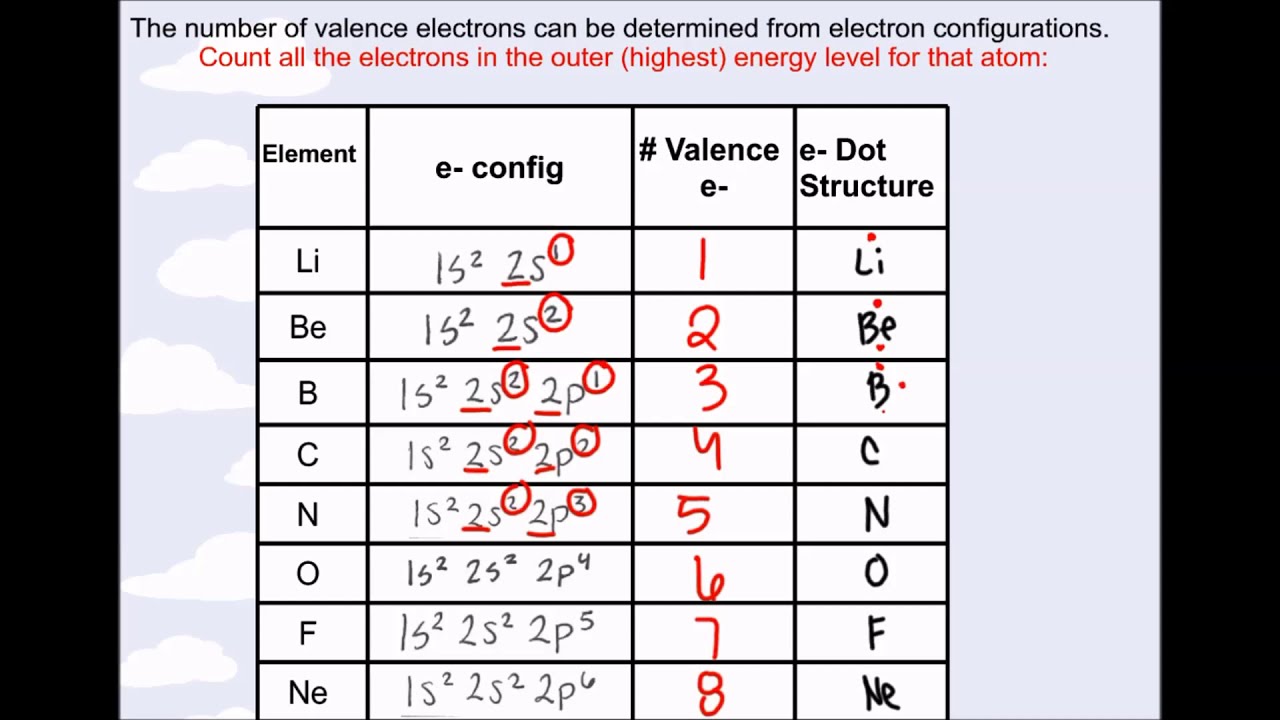
The two 4s electrons are in the highest principal quantum number, n 4, so they are the valence electrons. The path a specific element will take depends on where the electrons are in the atom and how many there are. Reason: The electron configuration of Fe is 1s2 2s2 2p6 3s2 3p6 4s2 3d5. In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is.\,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms. This will be how many valence electrons there are. Counting along the row/period, count the number of boxes to your element. Locate the element on the periodic table. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. The valence electrons are found based on where the element is on the periodic table. For non-transition metals, we count to 8, but for transition metals, we count to 12. Looking at the orbitals explains how valence electrons work for transition metals. 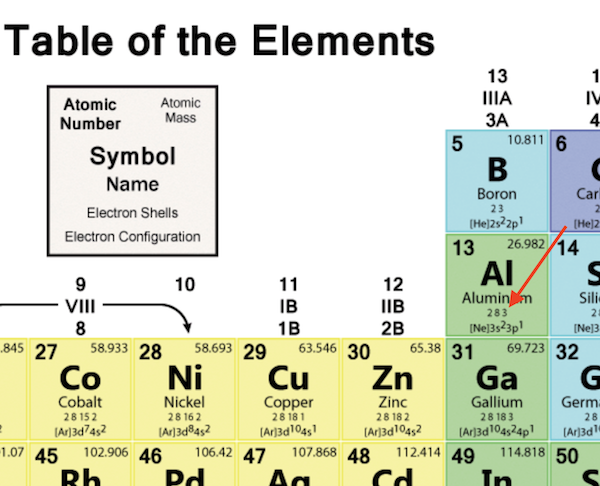
For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). The shells after (ignoring transition metals) represent the s and p-orbitals. So what does this have to do with our shells? The first "shell" represents the 1s orbital. For example, carbon has an electron configuration of 1s 22s 22p 2. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. It is said to occupy orbitals in an atom. EXAMPLE 11.12 ENHANCED EXAMPLE How many valence electrons. Electrons are involved in the chemical bonding and reactions of the atom. 
The way we count our electrons is by moving from right to left, starting at the beginning of the table. number of valence electrons to be used in the structure by adding the number. F-orbitals start appearing in the lanthanides and actinides (the separated two rows). number of bonds an element has in a compound in this book. P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). valence electrons may also be represented by a series of dots. Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level.Has 5 subshells, each holding 2 electrons, for a total of 10 electrons An explanation and practice for finding the number of valence electrons for elements on the periodic table.Has 3 subshells, each holding 2 electrons, for a total of 6 electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
